A recent research featured in the October edition of the Journal of the American Medical Association presents a Dutch investigation that studied approximately 90,000 women who underwent the Papanicolau test.
The investigators state that the liquid-based cytology which represents an alternative to the traditional Papanicolau test provides no advantage in comparison with the latter in finding pre-cervical cancer symptoms or the actual disease.
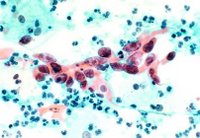
The medical world thought the Papanicolau test to be under the detection standards because it sometimes provided false- positive or negative results for the patients who underwent it. Regarding the usage of body cell entitled liquid-based cytology, the cells coming from the cervix are gathered thorough the use of a common sampling gadget and are put into a vial containing some preservation substance compared to the process of putting the sample on a glass slide, a thing which was used in the past. However, even if this represents a new detection method, various researches showed that the accuracy of the detection was not always a certain fact.
The team of scientists undergoing the investigation was led by Albertus Siebers from the Radboud University Nijmegen Medical Centre located in the Netherlands state Nijmegen. He and his colleagues started the research by comparing the screening accuracy of the Papanicolau tests and the liquid-based cytology. They looked at the positive results rate, the histological detection ones and the PPVs (positive predictive values). The investigation had a random character and comprised a number of 89,784 females who aged between 30 years and 60 years. All of the women were included in a screening process for cervical cancer which comprised 246 family Dutch clinics. From these family practices, 122 used liquid-based cytology and verified 49,222 women and the rest of the medical facilities used the traditional Papanicolau testing and verified 40,562 women. The females were controlled for CIN (cervical intraepithelial neoplasia) and were monitored for eighteen months until January. This took place in the year 2008.
The group of investigators discovered that the adjusted detection rate for cervical intraepithelial neoplasia garde 1+ was of 1.01, for the cervical intraepithelial neoplasia grade 2+ it was 1, for cervical intraepithelial neoplasia grade 3+ it was 1.05 and for carcinoma, the ratio was 1.69. As the investigators explain, this adjusted ratio did not vary very much from one for certain results of cervical intraepithelial neoplasia in the cases of some cytological call-off.
Due to the random character of the investigation, it can be thought that the propensity of cervical intraepithelial neoplasia was the same for both of the tested samples. This is why the inexistence of variation in the detection ratios and the positive predictive values during the specific research proved that the liquid-based cytology is not more sensitive or more accurate in discovering the cancer affecting the cervix compared to the traditional Papanicolau method.
The scientists concluded that there was not a significant variation in the positive rates provided by the cytological testing mechanisms. However, the evidence provided by this innovative detection process does not bring enough proves to state a similar accuracy in diagnosing women who suffer from cervical cancer. The analysis comprising the intent to treat and the way in which doctors can do this showed that the liquid-based cytology was not better than the Papanicolau test in the matter of finding positive rates of the already studied under the microscope results.
The same outcome came from the positive predictive values. In conclusion, the evidence provided by the investigation states that if the cytological testing is done in a very organized and qualitative screening for cervical cancer process, it does not differ from the traditional Papanicolau test.
Mark Schiffman and Diane Solomon, both medical doctors from the National Cancer Institute state their opinion regarding this research. Nowadays, being a doctor involved in the prevention of the cervical cancer seems to be a very challenging position. Even if the methods and tools used in the cervical detection process are updated and come in high numbers, no corroboration of vaccines, humanpapilloma virus tests, cytology, colposcopy and innovative ways would apply in the same way for all the women. They state that if we deal with areas around the Globe in which the health rates and quality of life are very high, the vaccines, liquid-based cytology or humanpapilloma virus tests could be a waste of resources if they are not taken out in a planned manner.
In addition, the medical doctors believe that the research undergone by Siebers team has made things simpler in the means that those persons involved in cervical cancer screening, monitoring and treating can use one of the two detection methods: the traditional Papanicolau test or the liquid-based cytology. Even if this decision-making process became more complex at the moment when the cytology appeared on the health market, the investigators managed to make the doctors see the exact role of this new detection method in applying treatments to their patients.
Labels: Cervical Cancer Research, Pap Smear, Pap Smear Test